described what happened to amount of product produced over time in the enzyme experiment.
Living cells and organisms could not function without enzyme controlled reactions. The more we sympathise well-nigh how enzymes function and the reactions they command, the better nosotros tin can use the machinery of nature to benefit man endeavours.
| Intro |  | Video |  | In focus |  | Podcast |  | Conclusion |  | Side by side Steps |  |
 How practise you measure the rate of enzyme controlled reactions?
How practise you measure the rate of enzyme controlled reactions?
Enzymes operate throughout biological organisms, both intracellularly and extracellularly. You lot volition be aware that enzymes are biological catalysts, pregnant they increment the rate of chemical reactions without undergoing any permanent change. Enzymes are made from long chains of amino acids, folded precisely into a three dimensional shape (or third structure) with an active site that allows it to operate as a catalyst. Any changes to this iii dimensional construction tin can change the shape of the active site and cause the enzyme to become denatured. This structure is represented in the lock and cardinal and induced-fit models of enzyme action, with the induced-fit model including the changes that can occur in enzyme shape to allow catalysis.
Given the range of enzyme controlled reactions, there is no single best method for measuring reaction rates equally the products of reactions vary profoundly. For example, catalase is a common intracellular enzyme that speeds the decomposition of hydrogen peroxide (a byproduct of metabolism) into h2o and oxygen. In this reaction the produced oxygen gas can exist collected and used as a way of measuring the reaction rate. Alternatively, the extracellular enzyme tripsin breaks down casein in milk, changing its color from white to clear. The reaction charge per unit can therefore be measured with a colorimeter, which will indicate the absorbance of light through the product. The spectrophotometer shown beneath is similar to a colorimeter, although it measures the manual, rather than the absorbtion of light.

As the dependent variable (the variable beingness tested) is the rate of reaction, we need to ensure that the measurements that we are taking are plotted against time. The independent variable (the variable we are manipulating, for case, enzyme concentration) could exist represented by plotting multiple lines on the same graph.
 What kinds of enzymes exercise researchers investigate?
What kinds of enzymes exercise researchers investigate?
 How practice you lot avoid errors?
How practice you lot avoid errors?
Errors can happen in even the best experiments, simply attention to detail and good experimental design can help to minimise both random and systematic errors.
Systematic errors ascend from either imperfections in the equipment existence used, or by improper technique in the laboratory. An example of a systematic fault would be if you were using a cuvette that was stained or scratched, then less light laissez passer would through your sample and all readings using that cuvette would exist affected. Similarly information technology is vital to properly make clean and dry out cuvettes, fill up them using a pipette, handle them only using gloves, and if possible, store them in a cuvette rack.

Random errors are most likely to occur because of the limitations of the equipment that you are using. For example, if your residuum is but accurate to a value of 0.1 grams but you need to measure out 250 milligrams of a substance. However, selecting the right tools for the correct job tin assistance minimise random errors. For example, an adjustable pipette will exist much amend at measuring out a few millilitres of a solution when performing a serial dilution than using a 50 mL beaker. If random errors are unavoidable due to equipment limitations, and so the all-time fashion to minimise them is to repeat the experiment as many times as possible to boilerplate out the error.
 Laboratory Confessions
Laboratory Confessions
In the Laboratory Confessions podcast researchers talk about their laboratory experiences in the context of A Level practical assessments. In this episode we look at the utilise of appropriate appliance to record quantitative measurements and the use of qualitative reagents to identify biological molecules.
 What can our measurements tell united states of america?
What can our measurements tell united states of america?
We can plot our results to assistance united states of america easily place the factors that can change enzyme activity. In that location is is a clear link here between the practical and theoretical elements of biological science equally the impact of concentration (of enzyme and substrate), inhibition, temperature and pH all accept feature effects on the rate of reaction plot.
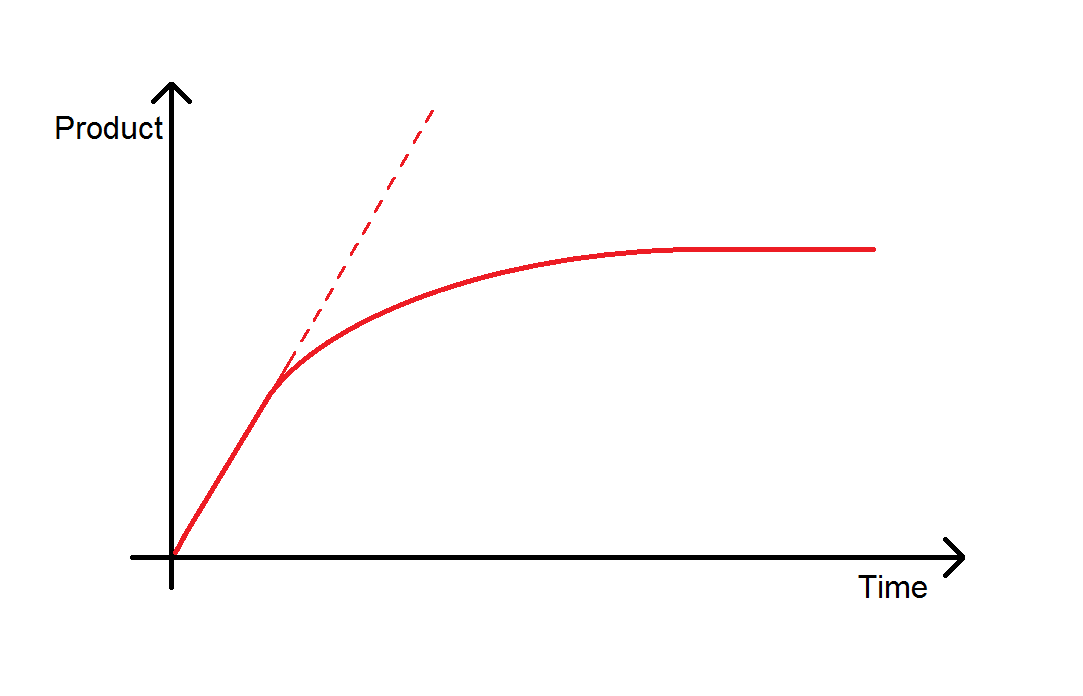
By plotting the amount of product against time, you lot should create a curve that looks a footling fleck like the one pictured. This plot is useful as it allows you to calculate the initial rate of reaction. The initial rate of reaction is the gradient of the straight line portion of the plot, shown by the dotted red line. The initial rate of reaction is when concentrations of enzyme and substrate are known, then this allows off-white comparison if yous then alter initial concentrations of enzymes or substrate.
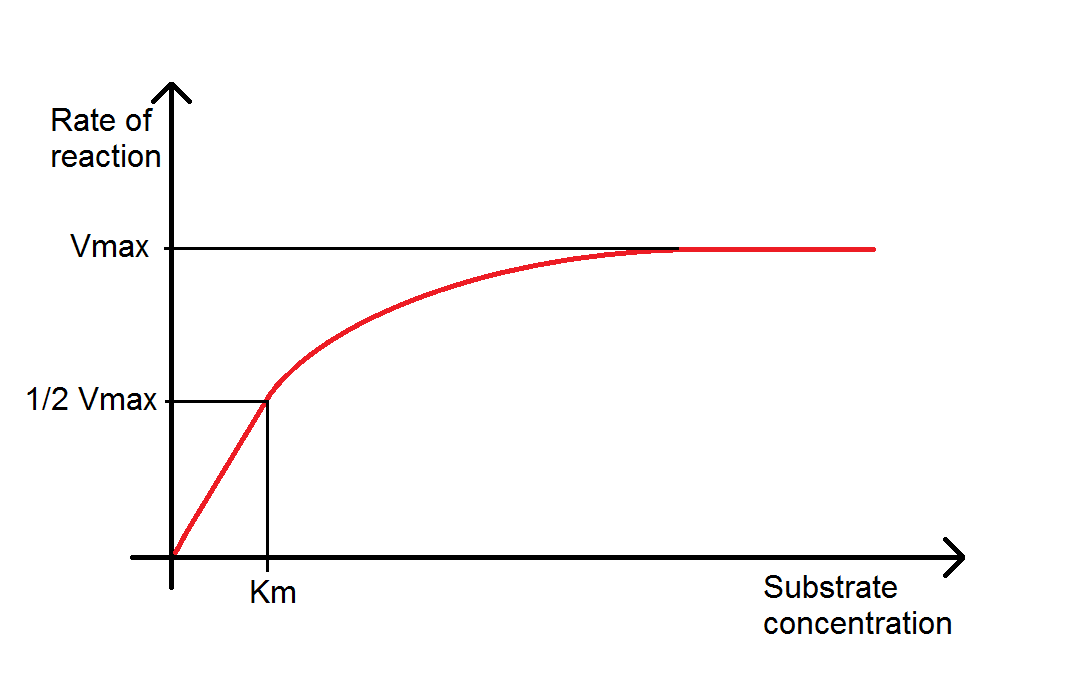
Once you have multiple reaction rates at different substrate or enzyme concentrations, it is then possible to accept this one step farther and plot reaction rate against substrate concentration, enzyme concentration, temperature or pH. Plotting reaction rate against substrate concentration typically gives a bend that is similar in shape to the product/time plot. It is, notwithstanding, a different bend and can tell you dissimilar things. Most importantly the Maximal Velocity (Vmax), which is when the enzyme is saturated with substrate and the charge per unit of reaction is highest, and the Michaelis-Mensten abiding (Km), which is a measure out of the enzyme's efficiency. Note that it is possible in some reactions for the reaction rate to drib once Vmax has been reached, every bit excess substrate can act as an inhibitor. A plot of reaction charge per unit against enzyme concentration will usually issue in a straight line, as typically the volumes of enzyme used are much lower than the volume of substrate; in other words it is similar to the directly line portion of the reaction rate/substrate plot. Eventually this plot will level off in a similar way to the reaction rate/substrate plot, although this is unlikely to be observed in classroom experiments! Reaction rate/pH plots should produce a classic bell curve, with the optimum pH at the elevation of the curve, and reaction rate/temperature plots should show an increasing rate of reaction with temperature until an optimum is reached (oftentimes betwixt 45 and 55 degrees Celsius), afterwards that the reaction rate drops off quickly as the enzymes become denatured.
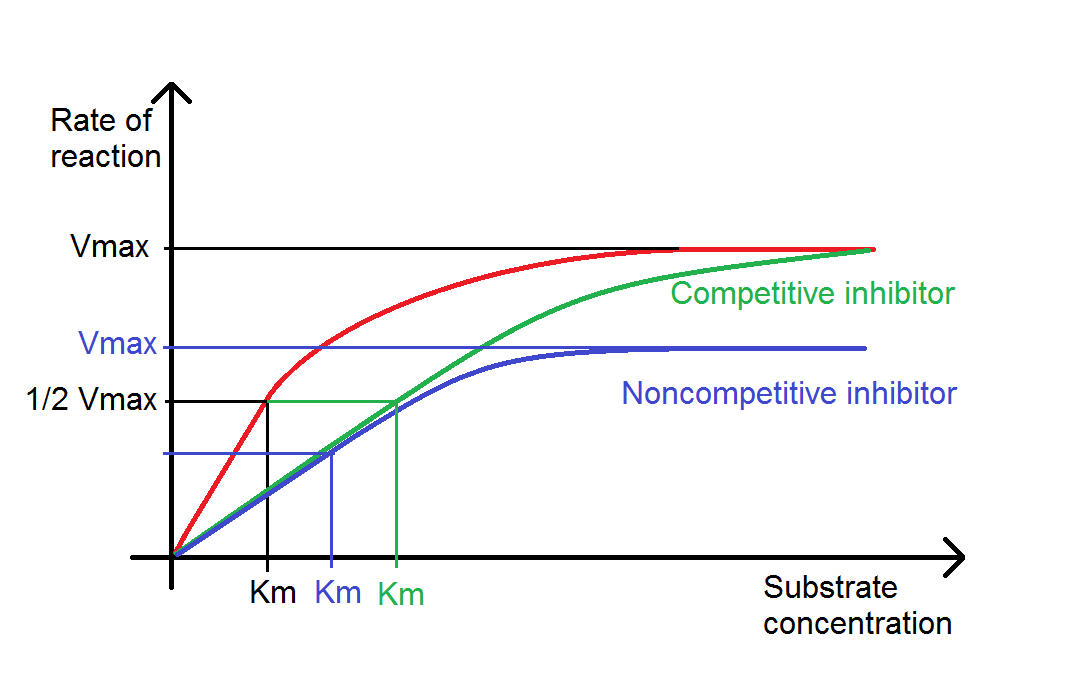
Dissimilar enzyme inhibitors will also modify reaction rate/substrate curves in unlike ways. A competitive inhibitor (for example, cyanide) competes with the substrate for the active site of the enzyme, reducing the rate of reaction at lower substrate concentrations. Given a high enough concentration of substrate the inhibitor tin be overcome, so the same Vmax equally the reaction without inhibition tin can be reached although the Km will be changed. Noncompetitive inhibitors (such as penicillin) do non use the agile site of the enzyme, possibly binding in some other identify and changing the conformational shape (an allosteric inhibitor). Increasing substrate concentration should still increase the reaction rate, but because enzymes can be inhibited regardless of how saturated their agile sites are, both the Vmax and the Km will exist changed.
 Adjacent steps...
Adjacent steps...
These links are being provided equally a convenience and for informational purposes only; they do not constitute an endorsement or an approval past the University of Birmingham of any of the data independent on external website. The University of Birmingham bears no responsibility for the accuracy, legality or content of the external site or for that of subsequent links. Please contact the external site for answers to questions regarding its content.
Source: https://www.birmingham.ac.uk/teachers/study-resources/stem/biology/stem-legacy-enzymes.aspx
0 Response to "described what happened to amount of product produced over time in the enzyme experiment."
Post a Comment